beam of light should be considered as an assembly of "units", each or which [using light frequency ( ν.
#Heisenberg principle free
he exchange of momentum between free electrons and radiation is very similar to the exchange.

For example, it means that a quantum particle does not move along a well-defined path through space.The smearing of position and momentum leads to an inherent indeterminism in the behaviour of quantum systems.he experimenter may fire an electron at a target and find that it scatters to the left, then, on repeating.
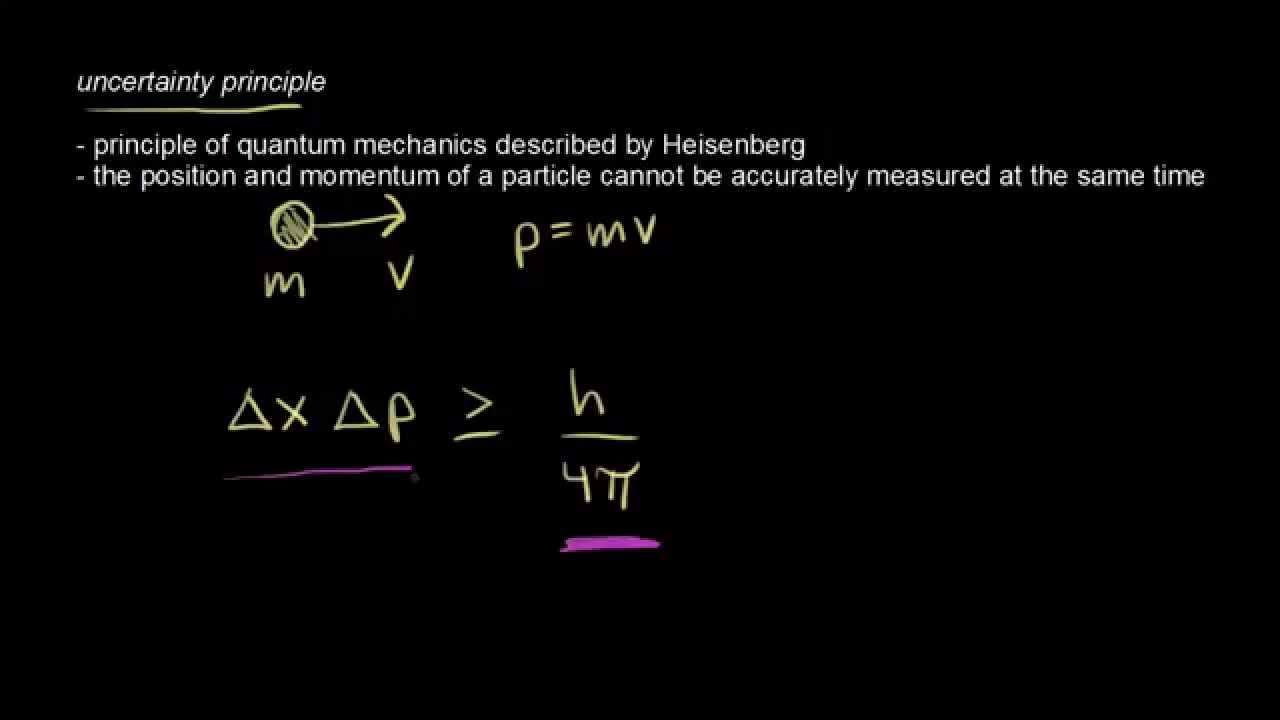
Heisenberg utilized such an observer effect at the quantum level as a physical "explanation" of quantum uncertainty. Historically, the uncertainty principle has been confused with a somewhat similar effect in physics, called the observer effect, which notes that measurements of certain systems cannot be made without affecting the systems, that is, without changing something in a system. The uncertainty principle, also known as Heisenberg's uncertainty principle in quantum mechanics, is any of a variety of mathematical inequalities asserting a fundamental limit to the precision with which certain pairs of physical properties of a particle known as complementary variables, such as position x and momentum p, can be known simultaneously. The spreading of the wave function in all directions shows that the initial momentum has a spread of values, unmodified in time while the spread in position increases in time: as a result, the uncertainty Δx Δp increases in time.

The evolution of an initially very localized gaussian wave function of a free particle in two-dimensional space, with colour and intensity indicating phase and amplitude.
